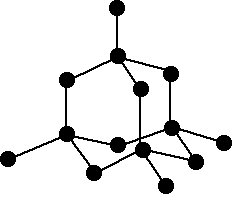
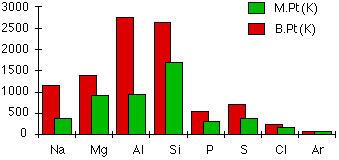
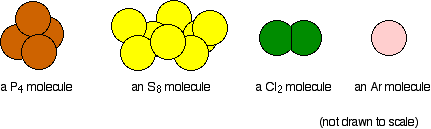Atomic and Physical Properties of Period 3 Elements
This page describes and explains the trends in atomic and physical properties of the Period 3 elements from sodium to argon. It covers ionisation energy, atomic radius, electronegativity, electrical conductivity, melting point and boiling point.
These topics are covered in various places elsewhere on the site and this page simply brings everything together – with links to the original pages if you need more information about particular points.
Atomic Properties
Electronic Structures
In Period 3 of the Periodic Table, the 3s and 3p orbitals are filling with electrons. Just as a reminder, the shortened versions of the electronic structures for the eight elements are:
| Na | [Ne]3s^1 |
| Mg | [Ne]3s^2 |
| Al | [Ne]3s^23p_x^1 |
| Si | [Ne]3s^23p_x^13p_y^1 |
| P | [Ne]3s^23p_x^13p_y^13p_z^1 |
| S | [Ne]3s^23p_x^23p_y^13p_z^1 |
| Cl | [Ne]3s^23p_x^23p_y^23p_z^1 |
| Ar | [Ne]3s^23p_x^23p_y^23p_z^2 |
In each case, [Ne] represents the complete electronic structure of a neon atom (1s22s22p6).
Note: If you aren't happy about electronic structures, it is essential to follow this link before you go any further.
First Ionisation Energy
The first ionisation energy is the energy required to remove the most loosely held electron from one mole of gaseous atoms to produce 1 mole of gaseous ions each with a charge of 1+.
It is the energy needed to carry out this change per mole of X.
The pattern of first ionisation energies across Period 3
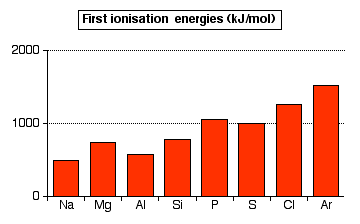
Notice that the general trend is upwards, but this is broken by falls between magnesium and aluminium, and between phosphorus and sulfur.
Explaining the pattern
First ionisation energy is governed by:
- the charge on the nucleus.
- the distance of the outer electron from the nucleus.
- the amount of screening by inner electrons.
- whether the electron is alone in an orbital or one of a pair.
Note: If you aren't certain about the reasons for any of these statements, you must go and read the page about ionisation energies before you go any further.
The upward trend
In the whole of period 3, the outer electrons are in 3-level orbitals. These are all the same sort of distances from the nucleus, and are screened by the same electrons in the first and second levels.
The major difference is the increasing number of protons in the nucleus as you go from sodium across to argon. That causes greater attraction between the nucleus and the electrons and so increases the ionisation energies.
In fact the increasing nuclear charge also drags the outer electrons in closer to the nucleus. That increases ionisation energies still more as you go across the period.
The fall at aluminium
You might expect the aluminium value to be more than the magnesium value because of the extra proton. Offsetting that is the fact that aluminium's outer electron is in a 3p orbital rather than a 3s.
The 3p electron is slightly more distant from the nucleus than the 3s, and partially screened by the 3s electrons as well as the inner electrons. Both of these factors offset the effect of the extra proton.
The fall at sulfur
As you go from phosphorus to sulfur, something extra must be offsetting the effect of the extra proton
The screening is identical in phosphorus and sulfur (from the inner electrons and, to some extent, from the 3s electrons), and the electron is being removed from an identical orbital.
The difference is that in the sulfur case the electron being removed is one of the 3px2 pair. The repulsion between the two electrons in the same orbital means that the electron is easier to remove than it would otherwise be.
Atomic Radius
The trend
The diagram shows how the atomic radius changes as you go across Period 3.

The figures used to construct this diagram are based on:
- metallic radii for Na, Mg and Al.
- covalent radii for Si, P, S and Cl.
- the van der Waals radius for Ar because it doesn't form any strong bonds.
It is fair to compare metallic and covalent radii because they are both being measured in tightly bonded circumstances. It isn't fair to compare these with a van der Waals radius, though.
The general trend towards smaller atoms across the period is NOT broken at argon. You aren't comparing like with like. The only safe thing to do is to ignore argon in the discussion which follows.
Note: If you aren't sure about the way that atomic radii are measured, it is essential to follow this link before you go any further.
Explaining the trend
A metallic or covalent radius is going to be a measure of the distance from the nucleus to the bonding pair of electrons. If you aren't sure about that, go back and follow the last link.
From sodium to chlorine, the bonding electrons are all in the 3-level, being screened by the electrons in the first and second levels. The increasing number of protons in the nucleus as you go across the period pulls the bonding electrons more tightly to it. The amount of screening is constant for all of these elements.
Note: You might possibly wonder why you don't get extra screening from the 3s electrons in the cases of the elements from aluminium to chlorine where the bonding involves the p electrons.
In each of these cases, before bonding happens, the existing s and p orbitals are reorganised (hybridised) into new orbitals of equal energy. When these atoms are bonded, there aren't any 3s electrons as such.
If you don't know about hybridisation, just ignore this comment – you won't need it for UK A-level purposes anyway.
Electronegativity
Electronegativity is a measure of the tendency of an atom to attract a bonding pair of electrons.
The Pauling scale is the most commonly used. Fluorine (the most electronegative element) is assigned a value of 4.0, and values range down to caesium and francium which are the least electronegative at 0.7.
The trend
The trend across Period 3 looks like this:
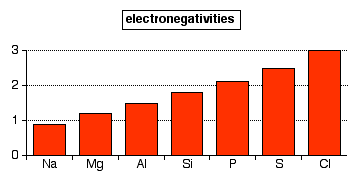
Notice that argon isn't included. Electronegativity is about the tendency of an atom to attract a bonding pair of electrons. Since argon doesn't form covalent bonds, you obviously can't assign it an electronegativity.
Explaining the trend
The trend is explained in exactly the same way as the trend in atomic radii.
As you go across the period, the bonding electrons are always in the same level – the 3-level. They are always being screened by the same inner electrons.
All that differs is the number of protons in the nucleus. As you go from sodium to chlorine, the number of protons steadily increases and so attracts the bonding pair more closely.
Note: If you want a more detailed discussion of electronegativity, follow this link to the bonding section of the site.
Physical Properties
This section is going to look at the electrical conductivity and the melting and boiling points of the elements. To understand these, you first have to understand the structure of each of the elements.
Structures of the Elements
The structures of the elements change as you go across the period. The first three are metallic, silicon is giant covalent, and the rest are simple molecules.
Three metallic structures
Sodium, magnesium and aluminium all have metallic structures.
In sodium, only one electron per atom is involved in the metallic bond – the single 3s electron. In magnesium, both of its outer electrons are involved, and in aluminium all three.